Ok, this is going to sound weird, but if my sons were ever going to ask to have an adult over for a playdate, I’m pretty sure they would chose either Thor, The Hulk, or Steve Spangler. It’s true, because, let’s face it, Steve Spangler is the superhero of science. He’s got the whole package . . . he’s smart, he’s funny, he’s got just the right amount of danger appeal and he’s messy. What more could a boy want in a pal?
THE IDEA
This week’s experiment started out when my littlest, MaGill, had been introduced to Pop Rocks by a friend. “What makes that popping sound in my ears when I eat Pop Rocks mom?” I was startled into a state of panic as my motherly instincts and childhood memories came colliding all at once. “HOLY MOLEY!! Tell me you didn’t drink soda with Pop Rocks! Your head could explode!” Or . . . just a second . . . maybe that was just some hogwash they told us kids when we were little to get us to quit eating those magnificent little exploding gems. Hmmm . . . I needed to catch my breath for a moment. What does make Pop Rocks POP? I set my 11 year old on the trail for the answer. He found it, and I found a fun experiment that’s going to be perfect for my kinder class this year! It’s McGuire boy tested and approved. ‘You HAVE to do this one with your class mom!’
(You can see the complete experiment laid out by Steve if you click on the button below.)
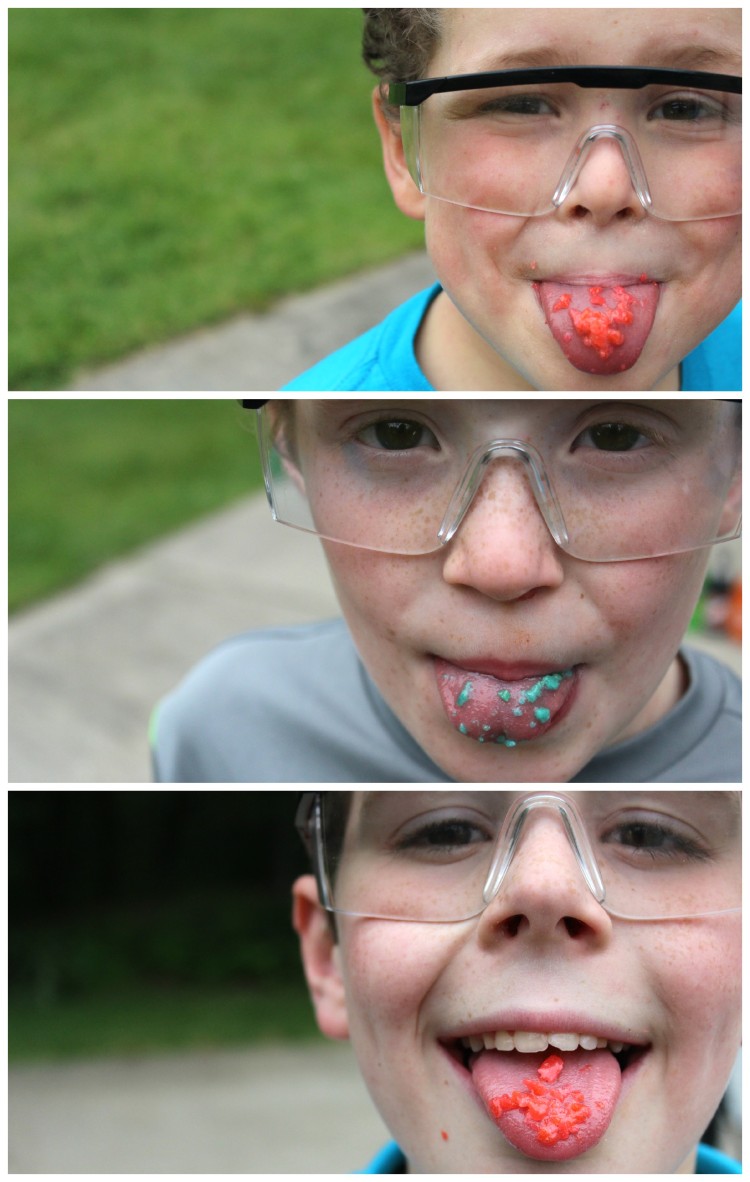
For three boys who seldom eat candy or get soda (We won’t count the days when there’s golf lessons and their instructor rewards them with the most holy unhealthy choices of soda and candy bars. What is he thinking?), the thought of getting their hands on soda AND candy for an experiment was too good to be true. They were all about being little Steve Spanglers for this one. Safety glasses on, and they’re ready to go.
THE DIFFERENTIATED CONNECTION
But, I know what you’re thinking, why would I ever do something like this is my kindergarten classroom? Well, let me tell you why and how it fits into differentiating. I think often, in our effort to ‘differentiate our instruction,’ we get very tied up in tiering. Tiering is JUST one way to differentiate, and we sometime completely forget that we need to differentiate in response to interest and learning profiles. A reluctant reader and writer is going to be much more inclined to write if he’s a science nut and you’re writing about a science experiment. How do I know this? I live with this reluctant reader/writer! But when it comes time to read the steps to an experiment or write about our hypothesis, he’s all about it. I have always said that differentiating in response to interest is the HOOK, and it really is. Don’t over-look it’s power to persuade even the most reluctant learners! You’ll be pleasantly surprised at the results.

WHAT YOU’RE GOING TO NEED
Your materials list for this one isn’t too lengthy. Here’s what you’re going to want to get:
- Safety glasses. We have these left over from our Nerf Gun party earlier this summer.
- A medium to large-sized balloon for each child. It needs to be big enough that it will fit over the rim of the soda top.
- a bottle of soda for each child. We don’t really drink soda, so I went to the Dollar Tree. The boys wanted to know if different sugar content or caffeine would change the results, so I picked up a variety or whatever they had.
- And, of course, Pop Rocks! I got mine again at the Dollar Tree, because you could get three in a packet for $1.00. That was the best deal I could find.
If you’re not familiar with Pop Rocks (What rock have you been living under?), they look like enlarged sugar granules. According to Steve, each little sample of rock candy has a small bit of pressurized carbon dioxide gas trapped inside. When that little bit of gas escapes the candy, it makes the POP that you hear.
Alone in your hand, of course, nothing happens . . .
. . . but when it hits your tongue and starts to dissolve, all kinds of popping occurs.
THE EXPERIMENT
So the question was, what would happen if we put this fabulous little carbon dioxide popping candy in another carbon dioxide releasing item such as SODA. How much carbon dioxide would be released and how much could we capture? Enough to fill a balloon? Enough to pop a balloon? Would the kind of soda used matter?
The first task we needed to complete was to get the entire packet of Pop Rocks into the mouth of the balloon. This was no small task and took adult ‘help’ which is why you’ll see no pictures of this part of the experiment. I will say, you may want to use a funnel to make it easier, but we were able to accomplish it with one person stretching the mouth of the balloon out while the other poured.
Once the balloons were filled, we carefully placed them over the rim of the soda, being careful not to let any of the rocks spill in . . . YET!
On a count of 1-2-3 . . . we all emptied our balloons into the soda.
The balloons quickly filled with the gas.
You could really see the rocks working . . .
The more they popped, the bigger the balloon would expand.
But then, we decided that we wanted to see how much bigger the balloons would get if we shook the soda. The gas really started working then.
It was a fun experiment. We actually tested out six different soda types and, despite our prediction that Mountain Dew would make the biggest balloon because it has the most sugar, it was Mr. Pibb and Diet Coke that had the best results. Of course, they want to test out other types of soda and do it again adding new variables to test such as heat and bottle types. But that will take another trip to the Dollar Tree.
This is going to be a great addition to my own classroom when we talk about gases. It might take parents donating a bottle of soda, but the Pop Rocks and balloons make it a very inexpensive hands-on demonstration of how gases are released. Plus, it’s cool and who doesn’t love Pop Rocks? Plus, you’re giving those kids with a high interest in science, some extra motivation and a hook for learning.
If you’d like to check out other cool science experiments, head on over to Steve Spangler’s page or check out my Pinterest board for other fun science experiments.
Follow Marsha McGuire (Differentiated Kindergarten)’s board Science on Pinterest.




















This is great! My students will love this–I’ve pinned it for next year. I love the stuff you share here on your blog; thanks so much.
Thanks so much Terri. I am so glad you like it.
What a wonderful experiment with gases!!!! I love it!
It was a ton of fun!
This is so great!! Looking forward to using it in the classroom. Thanks for blogging about these wonderful science experiments! And thanks to your boys too 🙂